|
Big Pieces or Small Pieces: Which React Faster?.(Projects listed in order of difficulty.) Students interested in projects involving chemical reactions may enjoy independent science projects like these: To create slime with specific properties, students use the engineering design process and also observe chemical reactions that occur as they use different materials and recipes.Ĭhemistry Projects about Chemical Reactions for Students Doing Independent Science Projects or Science Fair They then make additional batches of slime with different properties. Students learn to identify different properties of materials and then experiment with making their own slime using a basic recipe. In the How are New Materials Invented? video lesson, students investigate how materials are made and the role chemical reactions play. How are New Materials Invented? (Grade 5) Video Lessons to Explore Chemical Reactions 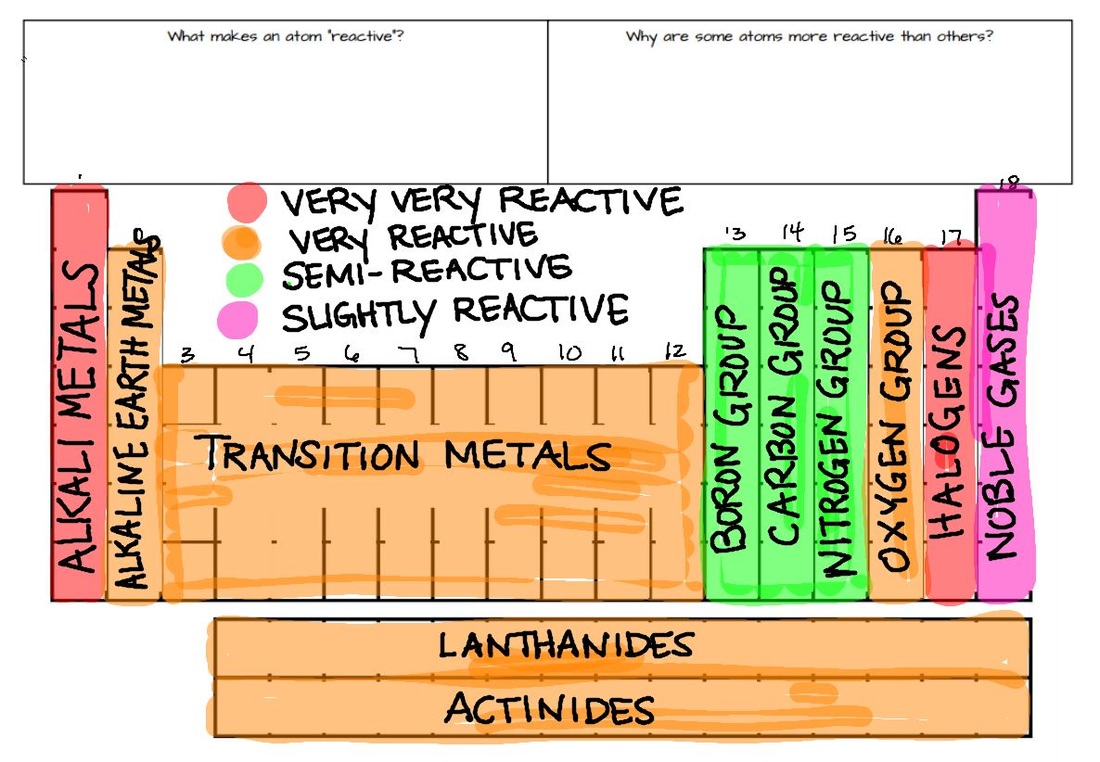
Note: for more information about these "types" of Science Buddies resources, see Understanding Science Buddies' Resources at the bottom of this resource. For students looking for science projects, we have also included a list of independent science and science fair projects. Resources are available in the following formats:Īt the bottom of this resource, you will find a list of key vocabulary words and additional related resources for teaching chemistry. Lessons, activities, and science demonstrations below include a range of foaming, fizzing, color-changing, and transforming experiments for teaching a unit on chemical reactions. Students also learn that the law of conservation of mass applies to all reactions and means that every atom of reactant can be accounted for as either product or waste when the reaction is complete. With classroom science experiments, activities, and independent student projects, students can learn about types of chemical reactions, including composition (also called synthesis or combination), decomposition, single replacement, double replacement, and combustion, and ways that the rate of a reaction can be sped up or slowed down. When teaching about chemical reactions, educators show chemical reactions in action and, when possible, let students get hands-on exploring reactions. * Some caveats: for example, aluminium will react slowly with water, if the thin aluminium oxide layer that prevents it from reacting is damaged.A chemical reaction occurs when two or more substances (reactants) are mixed together and result in one or more new substances (products). * This graphic doesn’t contain every metal in the periodic table. Majority of metals occur naturally in compounds, which we must remove them from. Some metals are so unreactive they occur largely uncombined with other elements, simple to obtain. This is because they can react with the compounds in metal ores, and displace the metals, aiding with their extraction. 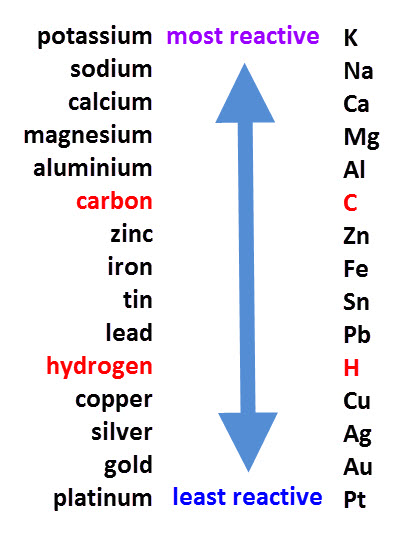
Carbon and hydrogen are also shoehorned in between entries in the list, despite being non-metals. Copper sulfate + zinc → zinc sulfate + copper Magnesium sulfate + zinc → NO REACTION The reactivity series also gives us an insight into why different metals are extracted from their ores in different ways. Conversely, if we react a metal with another metal lower in the series, no reaction will take place. The more reactive metal will take the place of the less reactive metal in the compound. If a metal compound reacts with a metal that’s above it in the reactivity series, a displacement reaction will occur. Uses of this: * predict the outcome of certain chemical reactions. Transition metals are much less reactive Gold and platinum have little in the way of chemical reaction. Closely followed by the marginally less reactive group 2 metals. Metals have a range of reactivities: Videos: the classic alkali metals in water demonstration The reactivity series offers a ranking of the metals in order of their reactivity. The text below has been excerpted from Compound Chemistry Student handout: Reactivity Series of Metals (PDF) This graphic places a selection of common metals into order of reactivity, as well as showing their reactions with air, water and steam.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
